Molecular structures
Posted on 18/05/2014
Contributed by

Helen Maynard-Casely

Helen Maynard-Casely
This is rocket science – ammonium perchlorate
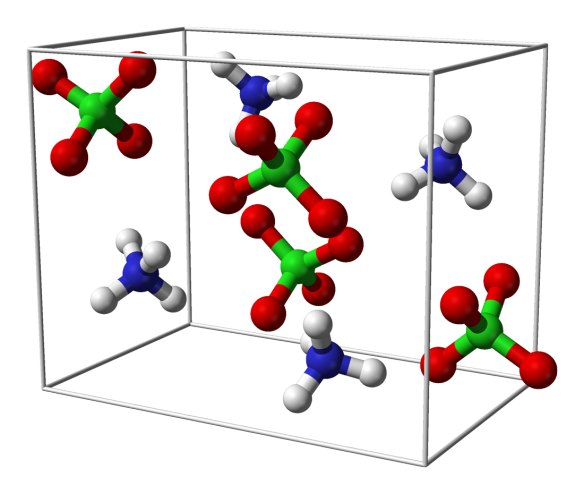
What does it look like?
What is it?
Though, when solid, this material is just a while powder, it is powerful enough to fuel rockets. Ammonia perchlorate forms the basis of a whole class of solid rocket fuels, known as ammonia perchlorate composite propellant (APCP). It can be molded and tightly packed into boosters and when ignited is a very powerful oxidiser, which is why it is so successful in this application. As a crystal structure it's actually a salt, as it is made up of tetragonal ClO4- anions balancing with NH4+ cations.
Where did the structure come from?
The structure of ammonium perchlorate was determined by Peyronel and Pignedoli in 1975.
Related articles
|
Saturating your food – Palmitic acid |
You must be taking the **** – Urea |
Important for this time of year – Vitamin C |