Molecular structures
Posted on 16/03/2014

Helen Maynard-Casely
Making a drug work smarter – Acemetacin co-crystals
What does it look like?
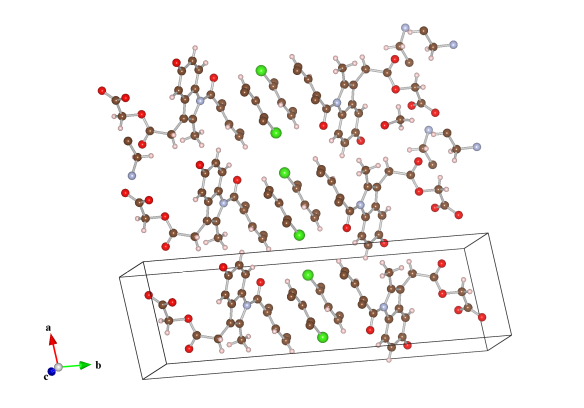
Image generated by the VESTA (Visualisation for Electronic and STructual analysis) software http://jp-minerals.org/vesta/en/
What is it?
Acemetacin is an anti-inflammatory drug that is used to treat chronic pain, especially for those suffering arthritis and pain following an operation. It is good for this application because, compared with other anti-inflammatory drugs, it doesn't cause as much damage to the stomach. But when mixed with water this drug will actually take in the water molecules into its structure, forming a less stable and not as useable hydrate form. So a recent investigation has looked at what other molecules will crystallise with acemetacin, to prevent it from looking to water to form a hydrate. What these investigators found was two molecules that formed 'co-crystals' with acemetacin. We've shown the co-crystal between acemetacin and piperazine. These co-crystals have an extra benefit in that they also have a greater solubility, meaning that the drug can get to work in your body faster.
Where did the structure come from?
This is a structure that has only been determined very recently and presented in the new open access journal IUCrJ.
Related articles
|
A tree bark suspension and a Nobel Prize later … We wonder less about the structural basis of the action of aspirin |
Sweet as – Glucose |
Perfect for the cold and 'flu season: Eucalyptus Oil |