Methane under pressure – the strange coincidence of methane B
Hello, Helen here – coordinator of the blog. As we look to the 'sprint finish' to the end of the year I thought I would write about a structure that I'm personally connected to – in fact it got me a PhD!
What does it look like?
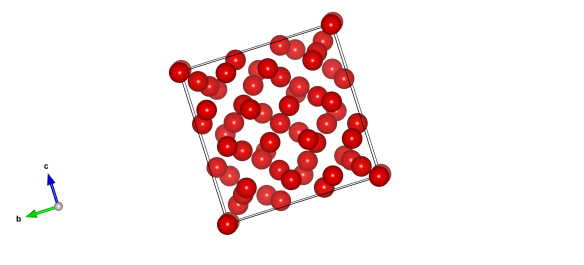
The structure of methane B. The red spheres are the carbon atoms found at the centre of the molecules. This was found with high-pressure X-ray synchrotron diffraction and unfortunately did not reveal the positions of the hydrogen atoms.
What is it?
Normally you only think of methane as a gas, but if you cool it to -145 °C it will liquefy, and then pretty soon after solidify to a solid that is quite like jelly (known a plastic solid – argon gas does the same thing).
But another way to freeze methane, like water too, is to compress it to very high-pressure - 5 GPa in fact (which is equivalent to five fully-grown African elephants standing on one stiletto heel). At this point methane freezes to a structure known as methane A, which we wrote about earlier this year. But if you keep squeezing methane, up to 8 GPa, then it undergoes a change to a new structure that, until yesterday, was a bit of a mystery.
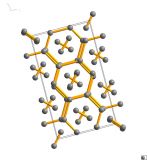
This form of methane is known as methane B, and is a rather complex cubic structure with 58 molecules needed in each repeating unit to describe it. It's pretty important as the pressure range that it is stable over extends to pressures that you would find inside the giant planets Uranus and Neptune. These wonders of our solar system are both thought to have an interior of mainly methane, water and ammonia, so knowing the structure of methane at these pressures really helps our understanding of them.
The rather strange thing about this structure is that we've seen it before – in manganese. Alpha-manganese also take up the same complex structure with 58 atoms all arranged the same. Not a pairing that you immediately put together, methane and manganese, but this is perhaps giving us a very vital clue as to how methane interacts at high-pressures.
Where did the structure come from?
This structure was published YESTERDAY in the Journal of Chemical Physics and was found, well, by me! The structure probably looks a little strange as I was only able to find the carbon atom positions (i.e. the centres of the methane molecules).
It's so shiny new that I've not got round to putting it into a database yet, but you can read the paper for free for the next 30 days! Though I promise I will put it into the Crystallography Open Database, first thing in the new year.