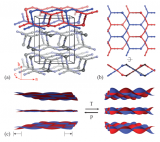
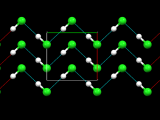
A big family of MOFs – Zeolitic imidazolate frameworks
What does it look like?
Picture generated with CrystalMaker (http://crystalmaker.com), from paper here: dx.doi.org/10.1039/c3cp54292k
What is it?
Zeolitic imidazolate frameworks (ZIFs) are a family of metal–organic frameworks, consisting of imidazolate linkers bridging metal centers with tetrahedral coordination (Zn, Co, Cd, Li, B, …). This tetrahedral arrangement is equivalent to the zeolite structures, inorganic porous structures that have wide industrial applications, with more than 200 framework geometries known to date. ZIFs couple this extremely wide structural variability with the possibility to feature functionalized imidazolate linkers (present in metal-organic frameworks in general). For these reasons, the ZIFs are the largest family of metal-organic frameworks known to date, with more than 100 different topologies synthesized experimentally, and many more predicted from quantum chemistry calculations.
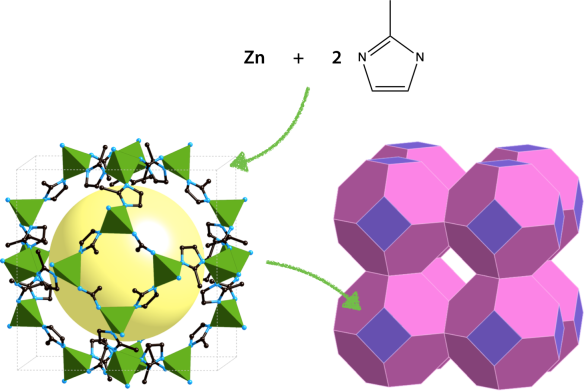
ZIF-8, the member of the family depicted above, has been one of the most studied. The Zn(imidazolate)4 tetrahedra are organized together to form cages (called "sodalite cages") with 8 hexagonal windows and 6 square windows. These cages are packed together to form the "sodalite" (or SOD) framework, from the name of the mineral where this topology was first encountered.
Like many members of the ZIF family, ZIF-8 is a very robust material compared with other metal-organic frameworks, with good chemical and thermal stability (up to 550°C). They have, on the other hand, relatively low stability under pressure.
Where did the structure come from?
References: dx.doi.org/10.1073/pnas.0602439103 and dx.doi.org/10.1002/anie.200503778