Negative Compression! Silver(I) tricyanomethanide
What does it look like?
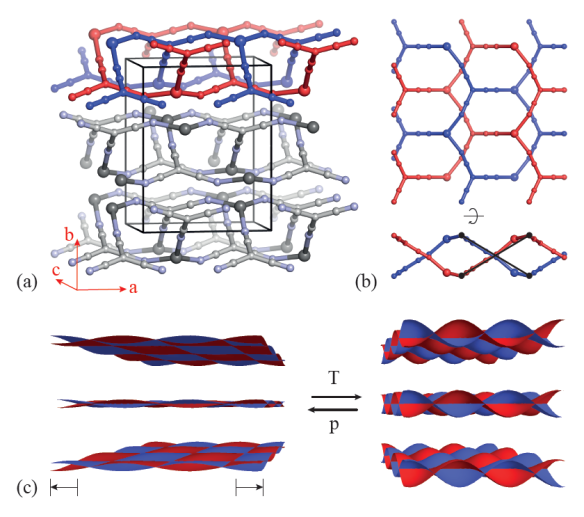
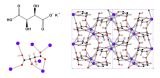
Silver(I) tricyanomethanide, Ag(tcm), has a layer-like network topology. In this network each Ag+ cation is coordinated to three tcm- anions in an approximately trigonal arrangement: likewise each tcm- anion is coordinated to three Ag+ centres. The resulting hexagonal (6,3) topology is sufficiently open that two honeycomb networks interpenetrate within each layer of the crystal structure. Adjacent layers interact via long Ag…N contacts that are sufficiently weak that the material behaves essentially as a two-dimensional framework.
What is it?
In the absence of a structural phase transition, it is a thermodynamic requirement that a system reduce its volume under hydrostatic pressure. The phenomenon of negative compressibility is the counterintuitive effect whereby this volume reduction couples to an expansion of the material in at least one linear dimension. In principle, the most extreme such response allowed thermodynamically is that of negative area compressibility (NAC), whereby a material expands along two orthogonal directions on increasing hydrostatic pressure. The remarkably few negative compressibility materials that have been characterised unambiguously expand only along one principal axis when compressed hydrostatically. The material Ag(tcm) exhibits both area negative thermal expansion and NAC due to a layer rippling mechanism illustrated in the figure above. The material therefore expands within the layers under hydrostatic pressure and contracts with increasing temperature. It is also noteworthy that the magnitude of NAC is almost as high as positive compressibility for conventional engineering materials.
Where does it come from?
Negative area compressibility in silver(I) tricyanomethanide. S. A. Hodgson, J. Adamson, S. J. Hunt, M. J. Cliffe, A. B. Cairns, A. L. Thompson, M. G. Tucker, N. P. Funnell and A. L. Goodwin, Chem Comm. 50, 5264-5266 (2014).
CCDC deposition numbers: 961474 – 961482