Iron trans-4,4'-azopyridine thiocyanate – Letting things down…
What does it look like?
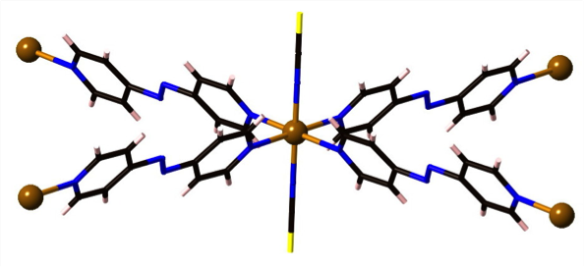
What is it?
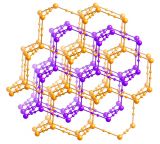
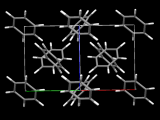
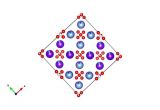
A feature of coordination polymers (or metal-organic frameworks – MOFs) is that many are porous, allowing the passage of gases and solvent molecules in and out of holes within their structures. An interesting feature of the coordination polymer shown here [Fe2(azopyridine)(NCS)2] is that those molecules coming in and out of the structure elicit a change in the magnetic properties and colour of the material.
It happens through a process known as spin-crossover. Magnetism is caused by unpaired electrons in atoms. Electrons have a 'spin', which can be either 'up' or 'down', and usually like to pair up with another electron in various orbitals around an atom. The two electrons in each orbital have opposite spin and thus cancel each other out. In some atoms, however, there are more orbitals available than pairs of electrons, and thus the electrons have a choice of spreading out amongst the orbitals as much as they can, or forming as many pairs as they can. The former case leads to a maximum number of unpaired electrons (and is known as the high spin state), and the later case has the fewest number of unpaired electrons possible, and is known as the low spin state. In spin crossover the atoms can swap between the high spin and low spin states when the temperature is changed, pressure is applied, or the environment around that atom is changed.
In the example shown above, there are iron atoms that are bridged by long linear ligands. Importantly, the iron atoms also have two short thiocyanate ligands on opposite sides. When the coordination polymer is made, it crystallises with ethanol molecules in the lattice that cause the thiocyanate ligands to lay over somewhat. In this state the material swaps from high spin to low spin as it is cooled, and the colour changes. When the ethanol is removed from the lattice, however, the thiocyanates become 'erect', standing up and bonding straight-on. This and other resulting movements in the lattice change the environment around the iron atoms enough to force them to stay high spin at all temperatures. Exposure of the material to ethanol vapour causes it to reabsorb the ethanol molecules, the thiocyanates flop over again, and it returns to the low spin state.
The fact that the presence or absence of ethanol in the structure has such a large effect on the thiocyanate geometry and hence the overall magnetic properties, particularly since they are not directly bonded to the iron atoms, is fascinating and has very interesting implications for tuning the guest responsiveness and signalling in this and other materials. It might also be the first example of "Brewer's Droop" on a molecular scale…
Where did the structure come from?
Source: "Guest-Dependent Spin Crossover in a Nanoporous Molecular Framework Material", G. J. Halder, C. J. Kepert, B. Moubaraki, K. S. Murray, J. D. Cashion, Science, 2002, 298, 1762. DOI: 10.1126/science.1075948
CCDC Deposition codes: 189340-189342