The Chinese puzzle molecule
What is it?
The Chinese Puzzle molecule is a molecular cation (a molecule with a positive charge – in this case +1) containing an inner cluster of "copper hydride" – 28 copper atoms (each with a +1 formal charge) and 15 hydrogen atoms (each with a -1 formal charge) enclosed in a shell of twelve dithiocarbamate (S2CR, R= C3H7) anions
What does it look like?
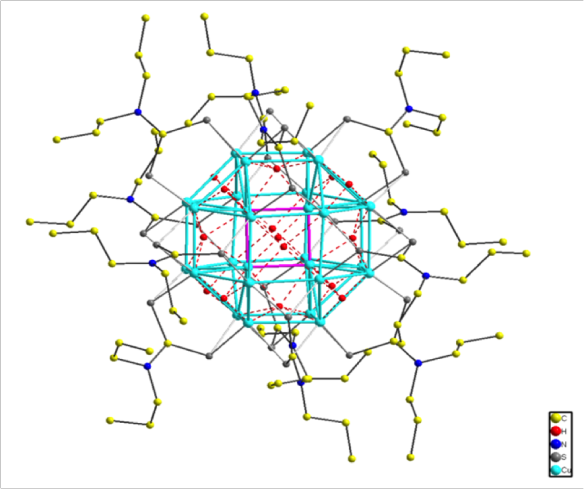
In this representation the 24 copper atoms making an outer rhombicuboctahedron (http://en.wikipedia.org/wiki/Rhombicuboctahedron) are shown in cyan and the inner four copper atoms are connected as though in a square shown in pink (these inner four copper atoms were the puzzle!). The hydrides are shown in red and the atoms of the dithiocarbamate are shown in grey/yellow/blue.
Where does it come from?
The compounds (many versions of this compound have been made with different types of dithiocarbamate) have been synthesized in the Laboratory of Professor Chenwei Liu of National Dong Hwa University, Taiwan and reported in Angewandte Chemie (http://onlinelibrary.wiley.com/doi/10.1002/ange.201403324/full) where it featured on the cover. The initial structure determination by X-ray diffraction revealed the presence of all the non-hydrogen atom sites, but interpretation of the electron density of the four inner copper sites was not trivial since it is complicated by disorder – the twelve sites which appear could be well modelled as either 2/3rd occupied sulfur or 1/3rd occupied copper. Chemical and spectroscopic analysis and neutron diffraction reveal that the 12 sites are in fact 1/3rd occupied copper. Initial spectroscopic analysis had also revealed the presence of eight hydrides which led to the study of the molecule by neutron diffraction by Alison Edwards using the KOALA instrument at ANSTO which revealed that it actually contains fifteen hydrides.
The inner core of the structure was very puzzling because the twelve sites that the copper atoms are distributed over have to be occupied in a physically and chemically reasonable manner – and it was hard to work out how this could be. The square shown in the figure above may be a small part of the answer, but the most likely model has the copper atoms distributed over a tetrahedron. We know from the crystallography that the 12 x 1/3rd copper sites are equally occupied and we know from nuclear magnetic resonance that the hydrides experience an averaged metal atom environment – there are three kinds in the NMR spectrum – one for each type of site.
The core of the structure has one hydride at the centre, surrounded by 4 copper atoms (which are disordered over the 12 sites of a cuboctahedron); this is surrounded by an octahedron of hydrides which is surrounded by the rhombicuboctahedron of copper and then a cube of hydrides, and finally the sulfur atoms from the dithiocarbamate form a truncated octahedron.
In the animation we look into the structure to see how the cuboctahedron of 12 x 1/3rd occupied sites can be reduced to 3 pairs of tetrahedra and then how the tetrahedra can reorient inside the cluster – changing which hydrides and outer copper atoms it connects to. This is a possible explanation of how the spectroscopic and crystallographic findings can co-exist – there is some kind of dynamic process occurring which results in the observed average crystallographic structure giving only 3 different signals for hydride in the NMR. If the tetrahedron of copper was fixed inside the cluster the hydrides of the octahedron should give two signals in the NMR but we only see one.