A crystal sandwich – Vermiculite
What does it look like?
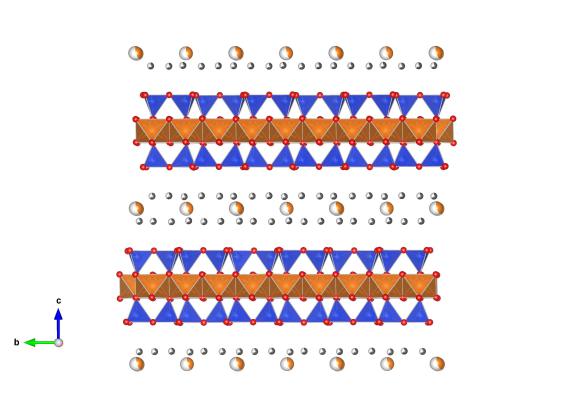
Image generated by the VESTA (Visualisation for Electronic and STructural analysis) software http://jp-minerals.org/vesta/en/
What is it?
Vermiculite is a layered silicate (also known as a phyllosilicates) mineral. It is very similar to Kaolinite in that respect but differs in the fact that it has an extra silicate layer (the blue tetrahedra) that sandwiches the layer of magnesium (in this case) and oxygen (which you can see as orange tetrahedra). These layers themselves then sandwich other elements (often water molecules with some small metals) that can sit in-between. This layered structure means that the structure of a vermiculite can swell up to take up elements from its surroundings, but can shrink at high temperatures when the water is expelled from the sandwich.
Where did the structure come from?
There's a lot of variation in the elements found in the vermiculite structure. This particular example is magnesium vermiculite, and is #9000146 in the Crystallography Open Database.