Glowing green – Willemite
What does it look like?
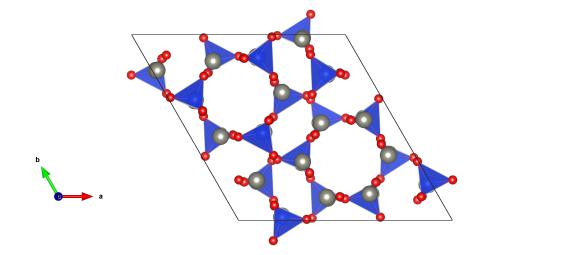
Light grey atoms are the zinc and the blue and red atoms make up the silicate units. Image generated by the VESTA (Visualisation for Electronic and STructural analysis) software http://jp-minerals.org/vesta/en/
What is it?
We’re having a few 'spooky' themed posts this week, kicked off with dry ice, so how about a green glowing mineral? Quite a number of minerals will 'glow-in-the-dark', mainly as a result of fluorescence after exposure to UV radiation.
Willemite is a zinc mineral, which fluoresces green because of manganese impurities within the crystal structure. This effect is strong enough that artificial Willemite was used in developing the first fluorescent lamps. Willemite is usually produced as an alteration from sphalerite, and is named after William I of the Netherlands.
Where did the structure come from?
The structure of willemite was found by Simonov et al. in 1977 – and its crystal structure can be found in the American Mineralogist Crystal Structure Database.