Biominerals #4 – The wonderful world of silica
What does it look like?
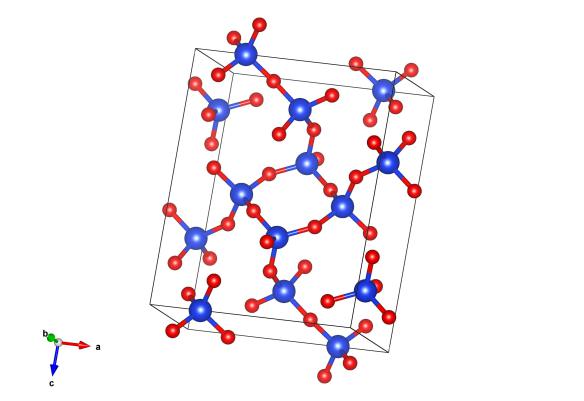
Blue atoms are silicon and red are oxygen. Image generated by the VESTA (Visualisation for Electronic and STructural analysis) software http://jp-minerals.org/vesta/en/
What is it?
One of the hallmarks of biomineralization is the elaborate morphologies that can be formed by creating minerals by these processes. This is nowhere more evident than in the exoskeletons of diatoms, single celled organisms that live in the seas all over the world. These exoskeletons are made from silica or silicon dioxide, often in its glassy or hydrated form. In fact, in Victorian times a number of people would arrange these tiny forms under a microscope to make beautiful patterns. Some people are still doing this today.
Most silica on the earth has the crystalline form of quartz, which we've covered a couple of times on the blog (see here and here), mainly because it is so important. But silica has a number of polymorphs (or different crystalline forms) that it can exist in, including moganite, which we're featuring here today. Moganite is quite an unusual form of silica because it is very low symmetry, monoclinic, and a very different arrangement of the silica units. For a long time there was controversy as to whether it existed or not!
Where did the structure come from?
The structure of moganite is #9005115 in the Crystallography Open Database.