What is the Rutile structure?
What does it look like?
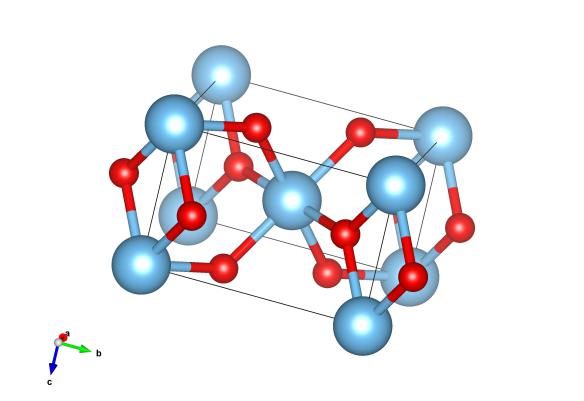
The crystal structure of rutile. Light blue atoms are the titanium and red are the oxygen. Image generated by the VESTA (Visualisation for Electronic and STructural Analysis) software http://jp-minerals.org/vesta/en/
What is it?
Rutile is another of those 'classic' crystal structures, in this case occurring in materials that have the general formula A2B, where all are bonded together. It's a bit different from many of the other common crystal structures as its basic building block isn't cubic. It is, in fact, a cell known as tetragonal, where although each of the corner angles are 90 degrees, one of the cell lengths can be different – making for an oblong box with square cross-section.
The 'original' rutile structure is actually titanium dioxide, or TiO2, and is a mineral commonly found in metamorphic rocks. It's a red mineral that often has white streaks in it. The white hints at one of the big uses for titanium dioxide as a white pigment: rutile is one of the main sources of the whitening part of our toothpastes!
Where did the structure come from?
An early study of the rutile mineral was conducted by W.H. Baur in 1956, and one of his later published structures is #9007531 in the Crystallography Open Database.