A crystal structure you eat with! Hydroxylapatite
What does it look like?
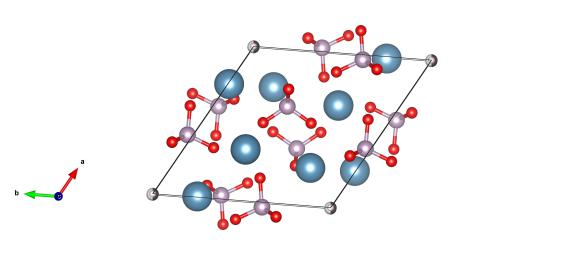
Red atoms are oxygen, light blue calcium, purple are phosphorus and the grey/white atoms are the hydroxyl positions. Image generated by the VESTA (Visualisation for Electronic and STructural analysis) software http://jp-minerals.org/vesta/en/
What is it?
This is a mineral that you make! Hydroxylapatite [Ca10(PO4)6(OH)2] is found in your teeth and bones, and many bone replacements (like hips) are covered in this mineral. It is one of only a handful of mineral (inorganic) materials that our bodies can make.
The enamel in our teeth is 96 % hydroxlyapatite, making it the hardest material in our bodies. However, once made we don't regenerate tooth enamel – repairing and maintaining this is what keeps dentists in business. When looking for suitable materials to repair tooth enamel with, the understanding of the crystal structure of hydoxlyapatite has been essential. In fact, a very new study has suggested that, triggered by small electrical currents, teeth can be made to re-mineralise and repair themselves.
Where did the structure come from?
The crystal structure of hydroxlyapatite was found by Kay, Young and Posner in 1964, using a combination of X-ray diffraction and neutron diffraction (to find the hydrogen atoms). The structure we've drawn up is #9001233 in the Crystallography Open Database.