Lacking in iron? – Iron (II) sulfate heptahydrate
What does it look like?
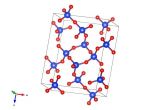
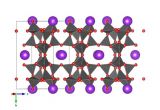
Image generated by the VESTA (Visualisation for Electronic and STructural analysis) software http://jp-minerals.org/vesta/en/
What is it?
Yesterday we featured vitamin C, so today we'd better follow it up with a mineral. We're told to maximise our vitamins and minerals in our diets – and generally the 'mineral' part refers to metal atoms that we get though eating things like green veg and sometimes even rubbed from our cutlery. But, for many reasons, we may sometimes be deficient in a mineral and need to take a supplement. For instance in iron supplements (which are taken when diagnosed with anaemia) the iron comes within an iron sulfate compound – often a hydrate. There are a few iron sulfate hydrate minerals that are found in nature, and similar to the source of iron found in the supplements we take. Today's structure is of melanterite, which is an iron sulfate heptahydrate (seven water molecules), a rare mineral found in Canada.
Where did the structure come from?
The structure of Melanterite can be found in the American Mineralogist Crystal Structure Database.