Seeing the New Year in red: the structure of strontium sitrate
What does it look like?
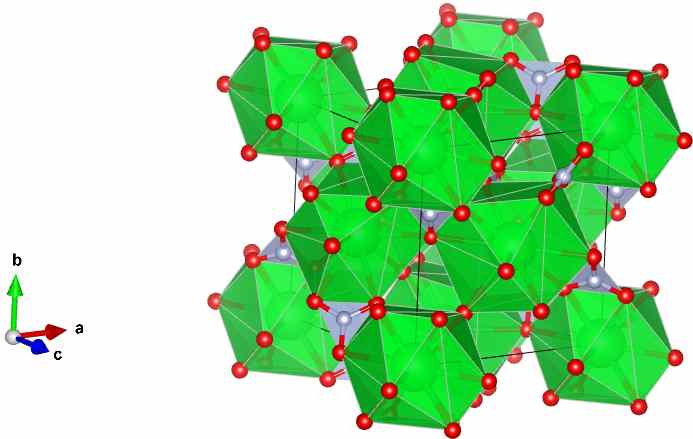
Image generated by the VESTA visualisation software. Green spheres are strontium atoms, red oxygen and lilac are nitrogen atoms.
As we all start to think about our hopes for the next year, throughout the world midnight on the 31st December will be heralded by fireworks. Bright explosives of colour have been a feature of our skies during celebrations for centuries, and there are several salt compounds that are used to make the bright colours we see. Here we focus on strontium nitrate salt – which is one of the family of strontium materials that give bright red colours to fireworks. It is the heat in the fireworks that sets off reactions that allow the electrons in the strontium atoms to excite and emit the red light. Not only beautiful in fireworks, strontium nitrate is also present in flares that are lit in emergency situations.

For more on the chemistry of fireworks – visit Compound Chem’s post.
Where did the structure come from?
The structure of Sr(NO3)2 shown was determined by Nowotny and Heger – and is published in Acta Crystallographica, Section C (1983), 39, 952-956. It is entry number 9011344 in the Crystallography Open Database.