Getting your cakes to rise – bicarbonate of soda
What does it look like?
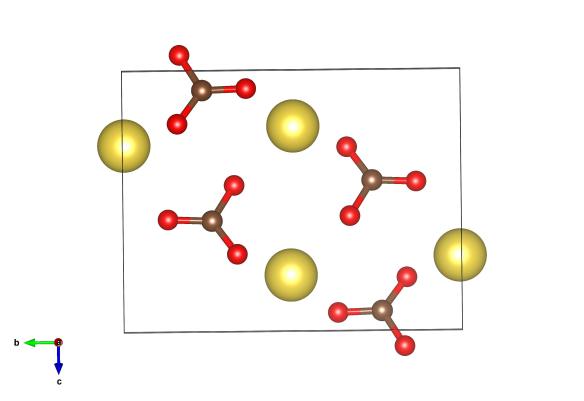
Yellow atoms are sodium, red are oxygen and the brown are carbon atoms. As this structure was found by X-ray diffraction no-one is quite sure where the hydrogen atoms are. Image generated by the VESTA (Visualisation for Electronic and STructural analysis) software http://jp-minerals.org/vesta/en/
What is it?
Raising agents (yeast, bicarbonate of soda and baking powder) work by adding carbon dioxide (CO2) gas to your mixture. This is produced as bubbles, which rise and add volume to your baking. Yeast works by a fermentation process; they eat the sugars in your bread and 'breathe' out carbon dioxide. But this is quite a slow process, which is why one has to leave bread to rise, and it is not always suitable for cakes and lighter baking. Bicarbonate of soda, sodium hydrogen carbonate (NaHCO3), is the quicker alternative for cakes. This releases carbon dioxide when reacted with something acidic (like fruit, buttermilk or honey). This acts straight away - that is why you can’t leave your baking batter for long before you put it in the oven.
But, if you’re baking something without the acidicness (i.e. chocolate chip cookies) you'll need to use baking powder. In your baking powder is an 'inbuilt' acid – usually cream of tartar (potassium hydrogen tartrate KC4H5O6), and when this released with moisture it reacts with the bicarbonate of soda to release the carbon dioxide.
Where did the structure come from?
The structure was first determined in 1933 by Zachariasen, but later modified (with better data) by Sass and Scheuerman in 1962. We've featured Sass and Scheuerman's structure of sodium bicarbonate but both can be found in the American Mineralogist Crystal Structure Database under the mineral name of sodium bicarbonate – Nahcolite