Superhard and superstrong – cubic Boron Nitride
What does it look like?
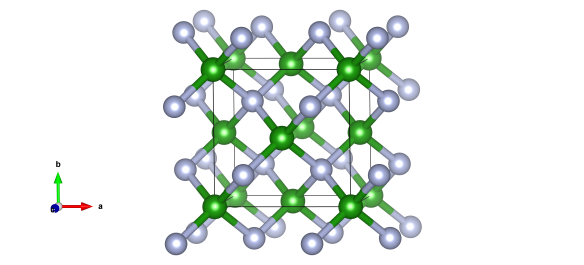
The green atoms are boron and the light blue are nitrogen. The light blue atoms are nitrogen and the pink ones are hydrogen. Image generated by the VESTA (Visualisation for Electronic and STructural Analysis) software http://jp-minerals.org/vesta/en/
What is it?
When you want a superhard material, diamond fits the bill pretty well every time. Not only is it super strong, but the benefit of being see-though means that if you're using a big gem you can see what you are compressing. However, diamonds are pretty expensive – so cheaper forms of superhard materials have been sought for quite a while.
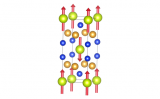
One of these is cubic boron nitride, made up of boron and nitrogen mixtures in a one-to-one mixture. If the structure is familiar, that's because this takes up the sphalerite or zinc blende structure. It is nearly as hard as diamond, and actually a bit more chemically stable.
Where did the structure come from?
Cubic boron nitride is #9008864 in the Crystallography Open Database.