From crystal to structure in 84 years
What is it?
In 1926 Jack Bean urease was the first protein to be crystallised1, earning a place in the history books for this humble plant protein and a Nobel Prize for James B. Sumner (see yesterday's post for more). Besides establishing that proteins could be crystallised, Sumner's work was remarkable for demonstrating that proteins could be isolated and purified whilst retaining enzymatic activity. Despite this landmark advance in protein biochemistry and crystallography, it was a further 84 years before the crystal structure of Jack Bean Urease was determined, in what must surely be the longest running crystallography endeavour in history2.
Where did the structure come from?
Urease is an enzyme in plants, bacteria and fungi that helps to break urea into ammonia and carbon dioxide. Whilst other urease structures were known – for example Helicobacter pylori urease, the structure of which helped to explain how this bacterium can survive in the acid environment of the human stomach3 – the structure of Jack Bean urease was only determined in 2010.
What does it look like?
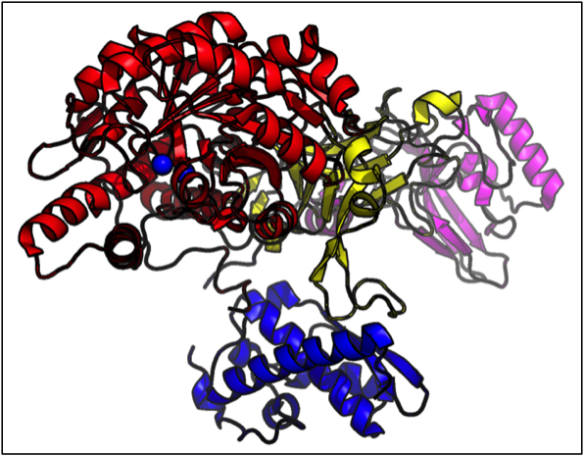
Plant ureases are single chain proteins. Jack Bean urease is a T-shaped molecule of four domains (coloured here in red, blue, yellow and pink). In common with other ureases, Jack Bean Urease binds two nickel ions (blue spheres) in its active site; these are required for enzymatic breakdown of urea.
This structure is Protein Data Bank ID 3LA4
References
- Sumner, 1926: J. Biol. Chem. 69:435-441
- Balasubramanian & Ponnuraj. 2010 J. Mol. Biol. 400:274-283
- Ha et al., 2001. Nature Structural Biology 8: 505-509