The Good and Bad of Prions: Proteins that cause Mad Cow disease
What is it?
Prion proteins play an important role in insulating our nerves – maintaining the protective myelin sheath that surrounds them.
However, prion proteins can adopt two states: a folded form and a misfolded form. The misfolded form is the 'bad' one: it can assemble into a tough fibril, suffocating the neurons in the brain, and after many years, it will eventually cause death.
The misfolded form of the prion protein has been implicated in many human pathologies from Alzheimer's disease to other neurodegenerative disorders.
This 'bad' form of prion is also the infectious agent in both Mad Cow disease and the human variant of the disease, Creutzfeldt–Jakob disease.
What does the 'good' form of Prion look like?
The correctly folded form of the prion protein is made up of alpha helices as shown in yellow and purple below. This structure was first published in Nature Structural Biology in 2001 (http://www.nature.com/nsmb/journal/v8/n9/full/nsb0901-770.html#f1)
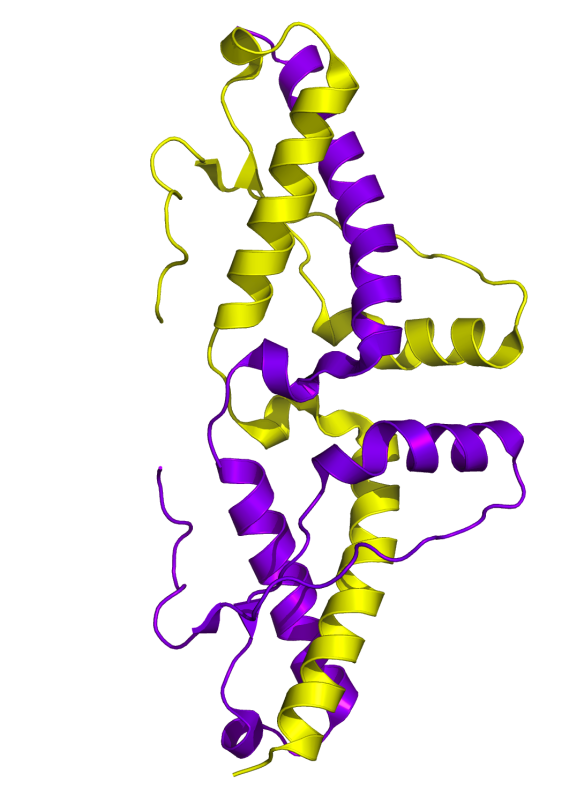
Image generated by Pymol (http://www.pymol.org/) using the coordinates from the Protein Data Bank (accession code: 1I4M).
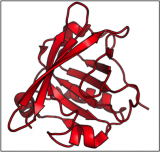
What does the 'bad' form of Prion look like?
When the prion protein misfolds, it can associate with other misfolded proteins, growing end to end to form a tough fibril. In 2008, we saw the first glimpse of how the misfolded protein can assemble together into this tough fibril that resembles something like an accordion.
You'll notice this form of the protein consists mostly of beta strands (shown as red arrows). The hydrophobic parts of the beta strands of this misfolded protein enable the proteins to 'velcro' together into these large fibrous structures. These fibrils cannot be broken down, and instead form large deposits in the brain.
This structure was published in Science in 2008 (http://www.sciencemag.org/content/319/5869/1523).
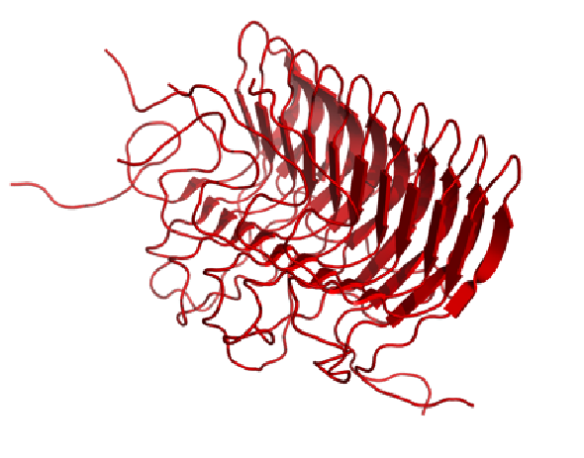
Image generated by Pymol (http://www.pymol.org/) using the coordinates from the Protein Data Bank (accession code: 2RNM).
'Bad' prions and Mad Cow disease
Mad Cow disease (also known as bovine spongiform encephalopathy) is a fatal disease that affects the central nervous system of adult cattle. It's called Mad Cow disease because the cow begins to act very strangely, eventually losing the ability to walk, and ultimately dies.
The misfolded form of the cow prion protein causes the disease. It is very unusual to have an infectious agent that is a protein and not a bacteria or a virus! But indeed, these misfolded prion proteins are highly infectious: just a small amount of a misfolded prion protein in the body can corrupt a whole bunch of surrounding 'good' prion proteins.
As the cow prion protein is very similar to the human prion protein, eating infected meat could mean that this Mad Cow disease spreads to humans. Unfortunately, prions are not destroyed even if the beef or material containing them is cooked or heat-treated. But the parts of the cow with the highest concentration of this protein (brain and spinal cord) can be removed to reduce the chance of disease spreading to humans. Fortunately, to date, very few people have been diagnosed with the human variant of the disease (Creutzfeldt–Jakob disease).
For more information about human spongiform encephalopathy and Mad Cow disease see: http://www.betterhealth.vic.gov.au/bhcv2/bhcarticles.nsf/pages/Creutzfeldt_Jakob_disease.