A Crystal Structure for the Perfect Steak: Papain
What does it look like?
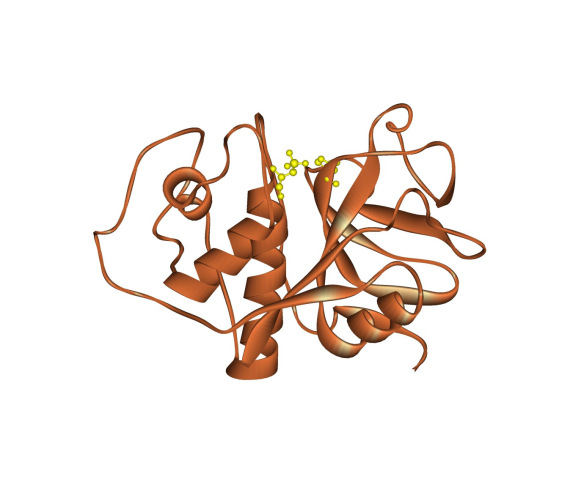
Papain from Carica papaya.
What is it?
The papaya fruit and pineapple are commonly used to make meat, particularly steak, tender. The reason for this is because papaya contains an enzyme called papain, which breaks down the tough fibrous material in meat to make it tender. Most powdered tenderisers will contain papain and pineapple also has similar enzymes called bromelain.
Papain is in the class of enzymes that breakdown proteins known as proteases. Papain is a cysteine protease because a cysteine amino acid (along with a histidine) is essential to papain's function of breaking the peptide bonds between the amino acids that make up a protein. In the image the essential cysteine (which is modified to a cysteinesulphonic acid to inactivate the papain so that the papain molecules don't break down each other during crystallisation) and histidine residues are shown in yellow to highlight the active site.
Apart from turning a tough piece of meat into quality steak papain also has a number of uses in medical research such as tissue culture and producing antibody fragments.
Where does it come from?
The X-ray crystal structure of papain was first solved by J. Drenth and colleagues in 1969 [1] and the structure shown is a later structure at higher resolution solved by I. G. Kamphius and colleagues in 1984 [2] (Protein Data Bank entry 9PAP).
[1] J. Drenth, J.N. Jansonius, R. Koekoek, H.M. Swen, B.G. Wothers (1969) Structure of papain. Nature 218: 929.
[2] I.G Kamphuis, K.H. Kalk, M.B. Swarte, J. Drenth (1984) Structure of papain at 1.65 Å resolution. Journal of Molecular Biology 179(2):233-256.