Now you've seen everything – superconductive concrete
What does it look like?
What is it?
In 2006 a senior scientist remarked in the journal Nature 'If that superconductor is made by doping concrete, I'll know it's time for me to retire'. We wonder if, the very next year, he was clearing his desk and getting his gold watch with the discovery of what happen when you dope today's crystal structure.
Dodecacalcium hepta-aluminate (12CaO.7Al2O3) – often just known as C12A7, is another of those useful crystal phases that crop up in cement, along with tricalcium aluminate. It even occurs in nature, as the mineral Mayenite. In the structure the calcium and aliuminium oxides form cages, which hold small amounts of oxygen ions. A group in Japan discovered that if you can replace the oxygen ions with electrons you can make the material behave like a metal, which is rather odd for concrete.
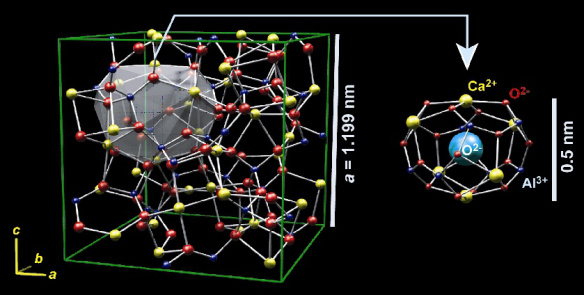
Crystal structure of C12A7. The cube is a unit cell. Two of the 12 baskets in the crystal contain oxygen ions. From http://techon.nikkeibp.co.jp/english/NEWS_EN/20070618/134409/
Then, in 2007, the cooled the doped material to 0.4 K and found that C12A7 becomes a superconductor! The key to this was how the electrons can move between the cages in the structure.
Where did the structure come from?
The crystal structure of Mayenite was first discovered by Buessem and Eitel in 1936, and is #1011034 in the Crystallography Open Database.